Figure 1 from Knowledge-data integration for temporal reasoning in a clinical trial system | Semantic Scholar
Therapeutics for Dengue: Recommendations for Design and Conduct of Early-Phase Clinical Trials | PLOS Neglected Tropical Diseases

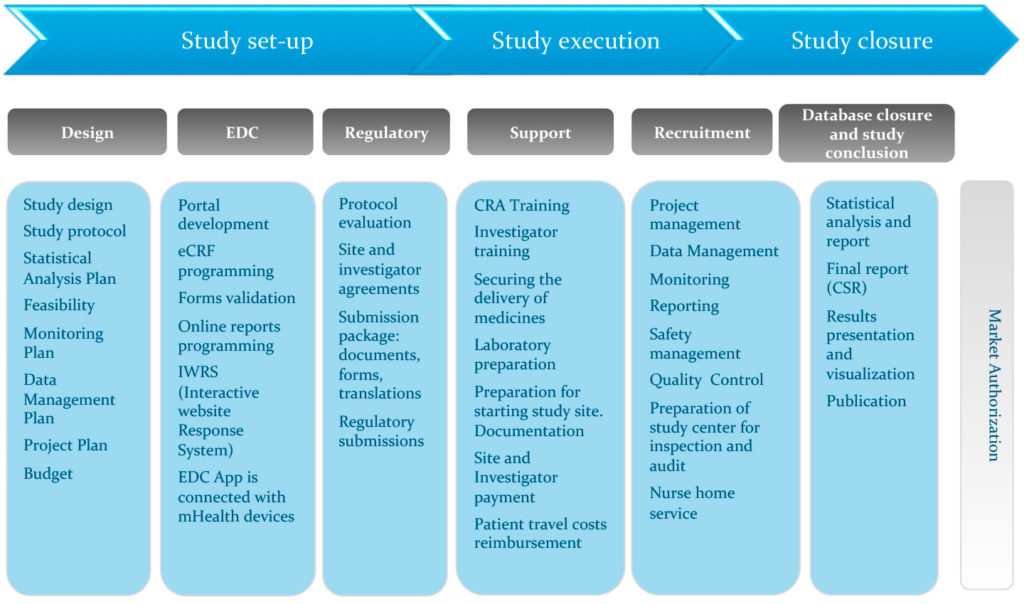
David Cloutier Director, Research Center Management and Development Budgeting for Industry Sponsored Clinical Trials. - ppt download

Single visit schedule throughout the trial. Patient in the embedded... | Download Scientific Diagram

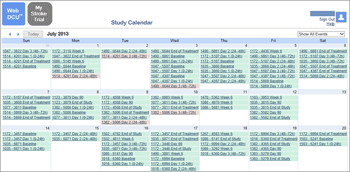
GitHub - RhoInc/participant-visit-listing: an interactive visualization of clinical trial schedule of events by participant
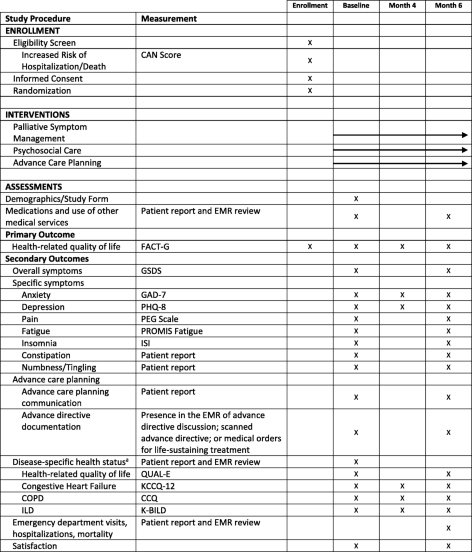
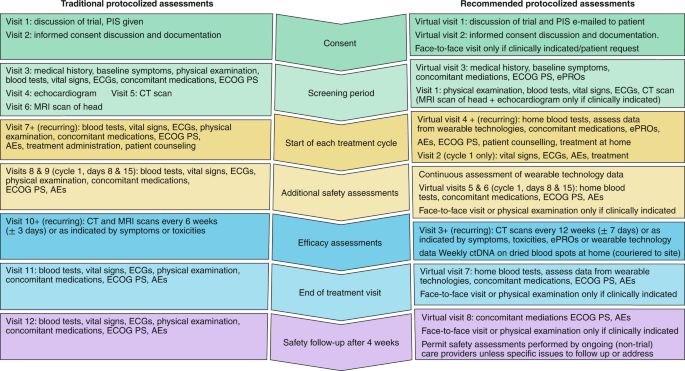
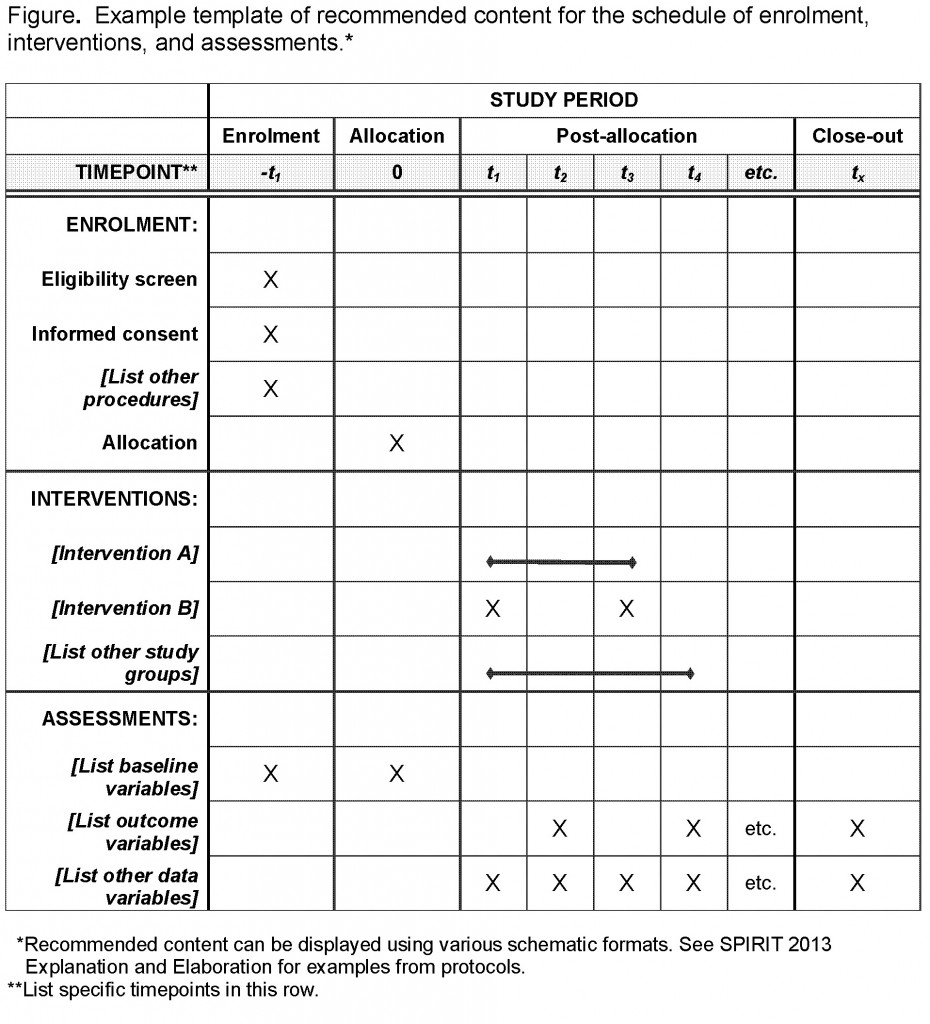
Advancing Symptom Alleviation with Palliative Treatment (ADAPT) trial to improve quality of life: a study protocol for a randomized clinical trial | Trials | Full Text

Schedule of the clinical trial. We randomly allocated the 80 patients... | Download Scientific Diagram

Table 1 from PCR adjusted cure rates in clinical trials of antimalarial drugs in Africa : Influence of extended follow-up and consecutive day blood sampling | Semantic Scholar